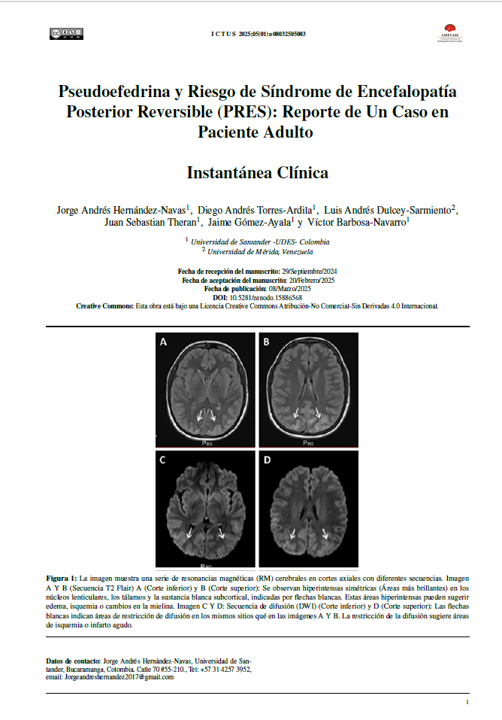
Abstract
Clinical Image on 'Pseudoephedrine and the Risk of Posterior Reversible Encephalopathy Syndrome (PRES): Case Report in an Adult Patient' presented in volume 5, Issue 1 of Ictus.
References
Glowacka K, Wiela-Hojenska A. Pseudoephedrine-benefits and risks. Int J Mol Sci. 2021;22(3):1150.
Torres MU, Delgado LV, Giraldo N, Urueña P, Franco S, Hernández OH. Síndrome de encefalopatía posterior reversible: reporte de un caso fatal y análisis de factores predictores de mal pronóstico. Biomédica. 2017;37(1):12–9.
Arama V, Ganea OA, Neagu D, Rosculet C, Arama SS. Update on the efficiency and safety of orally administered nasal decongestants. Rom J Infect Dis. 2023;26(4):125–34. Available from: https://rjid.com.ro/articles/2023.4/RJID_2023_4_Art-01.pdf
López Lois G, Gómez Carrasco JE. Reacción adversa por seudoefedrina: mioclonía inducida. An Pediatr (Barc). 2005;62(6):593–4.
Zerbib Y, Gibert L, Bennis Y, Masmoudi K, Maizel J, Brault C. Posterior reversible encephalopathy syndrome after self-medication with an oral decongestant: a case report. Front Med (Lausanne). 2022;9:843656.
Lizarazo J, Tibasosa D, Alandete S. Síndrome de encefalopatía posterior reversible. Acta Neurol Colomb. 2005;21(1):7–15.
Rodríguez Álvarez D, Molina Gutiérrez MA, Martínez Arias V. Intoxicación por antitusivos: a propósito de un caso. Rev Pediatr Aten Primaria. 2015;17(66):137–9.
Alzahrani Y. Pediatric posterior reversible encephalopathy syndrome: a review with emphasis on neuroimaging characteristics. Cureus. 2023;15(12):e50111.
Bartynski WS. Posterior reversible encephalopathy syndrome, part 1: fundamental imaging and clinical features. AJNR Am J Neuroradiol. 2008;29(6):1036–42.
Posterior reversible encephalopathy: beyond the original description. Neurología. 2015 [cited 2025 Jul 8]. Available from: https://www.neurologia.com/articulo/2015031020
Vernacchio L, Kelly JP, Kaufman DW, Mitchell AA. Medication use among children <12 years of age in the United States: results from the Slone Survey. Pediatrics. 2008;122(5):e1043–51.
Behar R. Anorexígenos: indicaciones e interacciones. Rev Chil Neuropsiquiatr. 2002;40(1):21–36.
Glowacka K, Wiela-Hojenska A. Pseudoephedrine—pharmacology and safety aspects. Int J Mol Sci. 2021;22(3):1150.
Martínez-Martínez F, Faus-Dáder MJ. Interacciones farmacológicas relevantes. Aten Primaria. 2001;28(9):625–34.
Salgado P, Pérez M, Sempere A. Beneficios clínicos de pseudoefedrina en rinitis alérgica. Farm Hosp. 2012;36(4):191–5.
Hinchey J, Chaves C, Appignani B, Breen J, Pao L, Wang A, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996;334(8):494–500.
Marra A, Vargas M, Striano P, Del Guercio L, Buonanno P, Servillo G. Posterior reversible encephalopathy syndrome: the endothelial hypotheses. Med Hypotheses. 2014;82(5):619–22.
Brinjikji W, Rabinstein AA, Lanzino G. Clinical and imaging features of posterior reversible encephalopathy syndrome. Clin Neuroradiol. 2012;22(2):113–20.
Ishikura K, Ikeda M, Hamasaki Y, Hataya H, Satomura K, Shimo T, et al. Posterior reversible encephalopathy syndrome in children: a nationwide survey in Japan. Pediatr Nephrol. 2013;28(6):1099–106.
National Institutes of Health. Pseudoephedrine: Drug information [Internet]. NIH Drug Database. Available from: https://www.nlm.nih.gov

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Copyright (c) 2025 Jorge Andrés Hernández-Navas; Diego Andrés Torres-Ardila (Autor); Luis Andres Dulcey-Sarmiento, Juan Sebastian Theran, Jaime Gómez-Ayala, Víctor Barbosa-Navarro